การแนะนำ
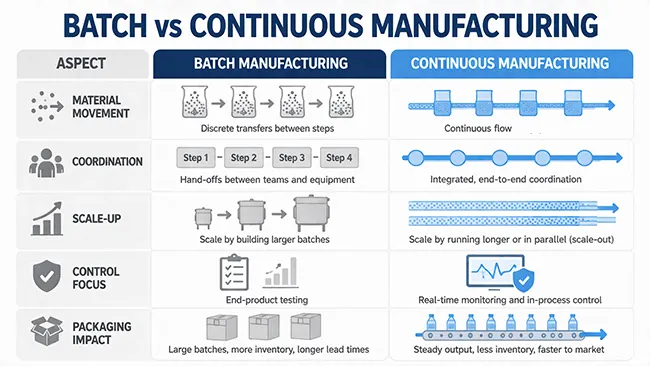
ในกระบวนการผลิตแบบต่อเนื่อง วัสดุจะเคลื่อนที่ผ่านขั้นตอนการผลิตที่เชื่อมโยงกันอย่างต่อเนื่อง แทนที่จะผลิตเป็นชุดๆ แยกกันอย่างชัดเจน ในระบบการผลิตแบบเป็นชุด วัสดุจะถูกแปรรูป เก็บรักษา และส่งต่อระหว่างขั้นตอนต่างๆ ในระบบการผลิตแบบต่อเนื่อง ขั้นตอนเหล่านั้นจะเชื่อมโยงกันอย่างใกล้ชิดมากขึ้น นั่นหมายความว่าสายการผลิตทั้งหมดขึ้นอยู่กับการไหลที่เสถียรจากขั้นตอนหนึ่งไปยังอีกขั้นตอนหนึ่ง
การเปลี่ยนแปลงนี้ส่งผลกระทบมากกว่าแค่ความเร็ว มันยังเปลี่ยนวิธีการรักษาสมดุลของการดำเนินงาน และวิธีการที่งานในขั้นตอนถัดไป เช่น การตรวจสอบ การบัฟเฟอร์ และการบรรจุภัณฑ์ ต้องสอดคล้องกับผลลัพธ์ในขั้นตอนก่อนหน้าด้วย
ยาเม็ดรับประทาน
ในการผลิต ควรคิดว่าการผลิตแบบต่อเนื่องเป็นโครงสร้างการผลิตที่แตกต่างออกไป ไม่ใช่แค่การเดินเครื่องจักรให้นานขึ้นเท่านั้น

การผลิตแบบต่อเนื่องในอุตสาหกรรมยาหมายความว่าอย่างไร
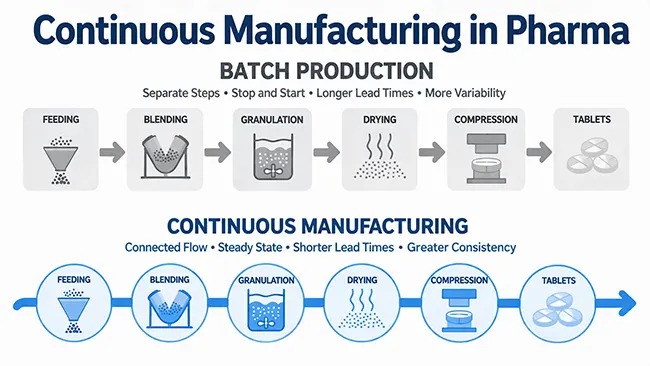
ลองนึกถึงการผลิตแบบต่อเนื่องว่าเป็นแบบจำลองการผลิตที่สร้างขึ้นบนพื้นฐานของการไหลเวียนที่เชื่อมโยงกัน วัสดุจะเคลื่อนที่ผ่านหน่วยปฏิบัติการที่เชื่อมโยงกันโดยมีช่วงหยุดพักระหว่างขั้นตอนหลักน้อยลง แทนที่จะมองแต่ละขั้นตอนเป็นบล็อกที่ค่อนข้างแยกจากกัน สายการผลิตจะถูกจัดเรียงเพื่อให้แต่ละขั้นตอนป้อนเข้าสู่ขั้นตอนถัดไปโดยตรงมากขึ้น
ในการผลิตแบบเป็นชุด วัสดุมักจะหยุดนิ่งระหว่างขั้นตอนต่างๆ ช่วงเวลาหยุดพักเหล่านั้นทำให้เกิดการแบ่งแยกในแต่ละกระบวนการ แต่ในการผลิตแบบต่อเนื่อง การแบ่งแยกนั้นจะลดลง เช่น การป้อนวัสดุ การผสม เป็นต้น
การทำให้เป็นเม็ด
กระบวนการอบแห้ง การบด และการอัดเม็ดยา จำเป็นต้องทำงานร่วมกันอย่างเป็นระบบมากขึ้น เป้าหมายเปลี่ยนจากการทำขั้นตอนใดขั้นตอนหนึ่งให้เสร็จสิ้นแล้วค่อยส่งต่อวัสดุในภายหลัง ไปเป็นการรักษาระดับการไหลของวัสดุให้คงที่เพียงพอเพื่อให้สายการผลิตทั้งหมดอยู่ในสมดุล
โมเดลนี้ให้ความสำคัญกับความสม่ำเสมอมากขึ้น การป้อนวัตถุดิบที่เสถียร การประสานงานระหว่างการดำเนินงาน และจังหวะการผลิตล้วนมีความสำคัญมากกว่า หากส่วนใดส่วนหนึ่งของสายการผลิตไม่เสถียร ผลกระทบจะส่งผลต่อขั้นตอนถัดไปได้เร็วกว่า เนื่องจากกระบวนการเชื่อมโยงกันอย่างแน่นหนากว่าการผลิตแบบเป็นชุด ดังนั้น การผลิตแบบต่อเนื่องจึงเปลี่ยนแปลงตรรกะการผลิตก่อนที่จะเปลี่ยนแปลงเครื่องจักรใดๆ
ด้วยเหตุนี้ หัวข้อนี้จึงขยายไปถึงเรื่องบรรจุภัณฑ์โดยธรรมชาติ กระบวนการผลิตต้นน้ำที่เชื่อมโยงกันมากขึ้นจะเปลี่ยนสิ่งที่อุปกรณ์ปลายน้ำต้องจัดการ ผลผลิตจากการอัดเม็ดยา เวลาในการตรวจสอบ ความจุของบัฟเฟอร์ และการจับคู่สายการบรรจุ จะง่ายขึ้นหรือยากขึ้นขึ้นอยู่กับความสม่ำเสมอของกระแสการไหลต้นน้ำ เมื่อคุณเข้าใจแนวคิดการผลิตอย่างชัดเจนแล้ว การพูดคุยเรื่องบรรจุภัณฑ์ก็จะเข้าใจง่ายขึ้นมาก
การผลิตแบบต่อเนื่องแตกต่างจากการผลิตแบบเป็นชุดอย่างไร?
ความแตกต่างหลักไม่ได้อยู่ที่ว่ารุ่นหนึ่งเก่าและอีกรุ่นหนึ่งใหม่ แต่อยู่ที่โครงสร้างของกระบวนการ การผลิตแบบเป็นชุดจะแบ่งการผลิตออกเป็นขั้นตอนแยกกัน โดยมีการหยุดพัก การขนย้าย และจุดพักระหว่างทาง ในขณะที่การผลิตแบบต่อเนื่องจะลดช่วงหยุดพักเหล่านั้นลง และมองสายการผลิตเหมือนรถไฟที่เชื่อมต่อกันเป็นหนึ่งเดียว
ความแตกต่างเชิงโครงสร้างนั้นทำให้สิ่งที่ผู้ปฏิบัติงานและวิศวกรให้ความสำคัญเปลี่ยนไป ในการผลิตแบบเป็นชุด คุณมักจะสามารถแก้ไขขั้นตอนหนึ่งได้ก่อนที่ขั้นตอนต่อไปจะเริ่มต้นขึ้น ในสายการผลิตแบบต่อเนื่อง ความผันแปรในขั้นตอนต้นน้ำจะเปลี่ยนแปลงไปอย่างรวดเร็ว ดังนั้นการควบคุมจึงขึ้นอยู่กับการรักษาเสถียรภาพของระบบโดยรวมมากกว่าการแก้ไขขั้นตอนใดขั้นตอนหนึ่งหลังจากที่เกิดขึ้นแล้ว
|
พื้นที่
|
การผลิตแบบเป็นชุด
|
การผลิตแบบต่อเนื่อง
|
|
การเคลื่อนย้ายวัสดุ
|
หยุด, รอ, โอน
|
การไหลเวียนที่เชื่อมต่อกันมากขึ้น
|
|
ความสัมพันธ์ระหว่างขั้นตอนต่างๆ
|
การแยกตัวที่มากขึ้น
|
การประสานงานที่ใกล้ชิดยิ่งขึ้น
|
|
ผลกระทบของการเปลี่ยนแปลงต้นน้ำ
|
มักจะล่าช้า
|
ไปถึงปลายน้ำได้เร็วกว่า
|
|
ตรรกะการขยายขนาด
|
ขั้นตอนเพิ่มเติมทีละขั้น
|
พึ่งพาการขยายขนาดแบบดั้งเดิมน้อยลง
|
|
ควบคุมการโฟกัส
|
การควบคุมระดับขั้น
|
ความเสถียรระดับเส้น
|
|
ผลกระทบของบรรจุภัณฑ์
|
สินค้าอาจมาถึงในปริมาณที่มากขึ้น
|
จังหวะการส่งออกมีความสำคัญมากกว่าสำหรับการจับคู่ในขั้นตอนถัดไป
|
ตารางด้านบนแสดงให้เห็นถึงความแตกต่างอย่างง่ายๆ นี่ไม่ใช่การจัดอันดับคุณภาพ แต่เป็นการเปรียบเทียบตรรกะของกระบวนการเท่านั้น ทั้งสองแบบสามารถใช้งานได้ เพียงแต่ต้องการสิ่งต่างๆ จากอุปกรณ์ กลยุทธ์การควบคุม และการวางแผนในขั้นตอนถัดไปที่แตกต่างกัน
เหตุใดผู้ผลิตยาจำนวนมากขึ้นจึงหันมาให้ความสนใจกับการผลิตแบบต่อเนื่อง?
ประการแรก การผลิตแบบต่อเนื่องสามารถลดความไม่効率บางประการที่เกิดขึ้นในการผลิตแบบหยุดและถ่ายโอนได้ ทั้งหน่วยงานกำกับดูแลและแนวทางปฏิบัติในอุตสาหกรรมต่างอธิบายว่าเป็นรูปแบบที่สามารถสนับสนุนการควบคุมกระบวนการที่แข็งแกร่งขึ้น ความผันแปรน้อยลง และในบางกรณี การขยายขนาดแบบดั้งเดิมระหว่างการพัฒนาและการผลิตเชิงพาณิชย์ก็ลดลงด้วย
นอกจากนี้ ยาเม็ดและแคปซูลก็เหมาะสมที่จะนำมาอภิปรายในเชิงปฏิบัติเช่นกัน ยาเม็ดและแคปซูลนั้นต้องอาศัยกระบวนการที่เชื่อมโยงกันหลายขั้นตอนอยู่แล้ว และการผลิตแบบต่อเนื่องทำให้กระบวนการเหล่านั้นชัดเจนยิ่งขึ้น ซึ่งทำให้เห็นประโยชน์ได้ง่ายขึ้น แต่ก็ทำให้เห็นความท้าทายได้ชัดเจนขึ้นเช่นกัน โดยเฉพาะอย่างยิ่งในเรื่องการป้อนวัตถุดิบ การปรับสมดุลสายการผลิต และการควบคุม
และนี่ไม่ใช่แค่แนวคิดในขั้นตอนการพัฒนาอีกต่อไปแล้ว การผลิตแบบต่อเนื่องนั้นเป็นที่ยอมรับมากพอที่จะมีความสำคัญในการวางแผนเชิงพาณิชย์ แต่ก็ยังใหม่พอที่หลายทีมกำลังพยายามหาว่ามันเหมาะสมกับสถานการณ์ใดและไม่เหมาะสมกับสถานการณ์ใด การผสมผสานเช่นนี้ทำให้มันเป็นหัวข้อที่มีประโยชน์สำหรับบริษัทต่างๆ ที่กำลังคิดเกี่ยวกับการออกแบบสายการผลิตในอนาคต การขยาย หรือการบูรณาการ
การผลิตแบบต่อเนื่องเปลี่ยนแปลงกระบวนการผลิตยาอย่างไร?
การให้อาหาร
การป้อนวัสดุมีความสำคัญมากขึ้น เพราะไม่ได้เป็นเพียงแค่การเริ่มต้นกระบวนการอีกต่อไป ในสายการผลิตแบบต่อเนื่อง อัตราการป้อนที่คงที่และคุณสมบัติของวัสดุที่สม่ำเสมอจะส่งผลต่อทุกส่วนในขั้นตอนถัดไป หากอัตราการป้อนเปลี่ยนแปลงมากเกินไป ส่วนที่เหลือของสายการผลิตจะมีพื้นที่น้อยลงในการรองรับการเปลี่ยนแปลงนั้น นั่นเป็นเหตุผลที่แนวทางสำหรับสายการผลิตแบบต่อเนื่องสำหรับยาเม็ดรับประทานเน้นย้ำถึงการป้อนวัสดุโดยวิธีชั่งน้ำหนัก (loss-in-weight feeding) ว่าเป็นกระบวนการที่แตกต่างและสำคัญ
การผสมและการทำเม็ด
การผสมและการบดเม็ดก็มีพฤติกรรมที่แตกต่างออกไปเมื่อเป็นส่วนหนึ่งของกระบวนการผลิตที่เชื่อมต่อกัน แทนที่จะเป็นขั้นตอนที่มีช่วงหยุดพักชัดเจน พวกมันจะกลายเป็นส่วนหนึ่งของระบบที่เคลื่อนไหว ซึ่งจะเพิ่มคุณค่าของพฤติกรรมของวัสดุที่คงที่มากขึ้นและการประสานงานที่ใกล้ชิดยิ่งขึ้นระหว่างขั้นตอนต่างๆ ในทางปฏิบัติ สายการผลิตจะขึ้นอยู่กับการรักษาสมดุลของกระบวนการมากกว่าการแก้ไขชุดผลิตภัณฑ์ที่เสร็จสมบูรณ์ชุดหนึ่งก่อนเริ่มชุดต่อไป
การอัดเม็ดยาและจังหวะการขับออก
การอัดเม็ดยาเป็นกระบวนการที่ผู้ผลิตหลายรายได้รับผลกระทบโดยตรงมากที่สุด
เครื่องอัดเม็ดแบบหมุน
ในระบบการผลิตแบบต่อเนื่อง เครื่องจักรไม่ได้เป็นเพียงส่วนหนึ่งของกระบวนการในขั้นตอนสุดท้ายเท่านั้น แต่กลายเป็นส่วนหนึ่งของสายการผลิตที่ต้องการวัตถุดิบจากต้นทางอย่างสม่ำเสมอ ดังนั้นจังหวะการผลิตจึงมีความสำคัญมากขึ้น เพราะประสิทธิภาพการอัดเม็ดยาจะส่งผลต่อความสอดคล้องของระบบโดยรวม รวมถึงการตรวจสอบและบรรจุภัณฑ์ในขั้นตอนต่อไปด้วย
การผลิตแบบต่อเนื่องเปลี่ยนแปลงบรรจุภัณฑ์ยาอย่างไร?
ประการแรก การจับคู่ผลผลิตเปลี่ยนแปลงไป เมื่อการผลิตยาเคลื่อนไปในทิศทางที่ราบรื่นและเชื่อมโยงกันมากขึ้น คุณจะไม่สามารถวางแผนในส่วนปลายน้ำได้
เภสัชกรรม
การจัดการบรรจุภัณฑ์โดยคำนึงถึงการส่งมอบที่ไม่สม่ำเสมอทำได้ง่ายเช่นกัน การสำรองสินค้า การจัดเก็บชั่วคราว และการปรับสมดุลสายการผลิตยังคงมีความสำคัญ แต่ในปัจจุบันสิ่งเหล่านี้ทำหน้าที่ในกระบวนการที่พยายามปกป้องการไหลเวียนของสินค้า แทนที่จะเป็นกระบวนการที่สร้างขึ้นโดยคำนึงถึงช่วงหยุดพักที่ยาวนานขึ้นระหว่างล็อตสินค้า
ประการที่สอง ตรรกะการตรวจสอบและการคัดแยกเปลี่ยนแปลงไป กระบวนการผลิตที่กระชับขึ้นหมายความว่าการควบคุมคุณภาพและการตัดสินใจในการจัดการต้องก้าวให้ทันกับผลผลิต นั่นไม่ได้หมายความว่าบรรจุภัณฑ์ทั้งหมดจะกลายเป็นแบบต่อเนื่องในลักษณะเดียวกันโดยทันที แต่หมายความว่าบรรจุภัณฑ์ต้องตอบสนองต่อสภาวะต้นน้ำได้เร็วและสม่ำเสมอยิ่งขึ้น เนื่องจากสายการผลิตมีการแบ่งส่วนน้อยกว่าในกระบวนการผลิตแบบเป็นชุด
ประการที่สาม การประสานงานระหว่าง
เครื่องบรรจุภัณฑ์แบบแผงพลาสติก
,
สายการนับเม็ดยาและการบรรจุขวด
, และ
เครื่องบรรจุกล่อง
การเปลี่ยนแปลง การผลิตแบบต่อเนื่องไม่ได้กำหนดรูปแบบบรรจุภัณฑ์โดยอัตโนมัติ แต่จะเปลี่ยนวิธีการที่รูปแบบนั้นต้องสอดคล้องกับผลผลิต สายการผลิตที่มีจังหวะการผลิตต้นน้ำที่สม่ำเสมอกว่า มักจะได้รับประโยชน์จากการวางแผนปลายน้ำที่มองการอัดเม็ด การตรวจสอบ การจัดเก็บ และการบรรจุเป็นลำดับการผลิตที่เชื่อมโยงกัน ไม่ใช่เป็นแผนกแยกต่างหากที่แก้ปัญหาที่แตกต่างกัน นี่คือบทเรียนเชิงปฏิบัติการจากแนวทางการผลิตแบบบูรณาการที่อธิบายไว้สำหรับสายการผลิตยาเม็ดรับประทาน
การผลิตแบบต่อเนื่องช่วยเพิ่มประสิทธิภาพการผลิตได้เสมอหรือไม่?
ไม่เสมอไป การผลิตแบบต่อเนื่องสามารถเพิ่มประสิทธิภาพได้ แต่ก็ต่อเมื่อสายการผลิตพร้อมสำหรับความต้องการที่มาจากการเชื่อมต่อกระบวนการที่แน่นแฟ้นยิ่งขึ้น หากการป้อนวัสดุไม่เสถียร คุณสมบัติของวัสดุเปลี่ยนแปลงมากเกินไป หรือการจัดการในขั้นตอนถัดไปไม่ทันการณ์ ประโยชน์ตามทฤษฎีของการไหลอาจกลายเป็นปัญหาการประสานงานได้อย่างรวดเร็ว
ดังนั้นอย่าสรุปแบบง่ายๆ ว่า “การผลิตแบบต่อเนื่องดีกว่า” มุมมองที่ถูกต้องกว่าคือ การผลิตแบบต่อเนื่องเปลี่ยนแหล่งที่มาของประสิทธิภาพ ในการผลิตแบบเป็นชุด คุณมักจะปรับปรุงประสิทธิภาพได้โดยการปรับปรุงแต่ละขั้นตอนให้เหมาะสมที่สุด แต่ในสายการผลิตแบบต่อเนื่อง ประสิทธิภาพขึ้นอยู่กับความสมดุลของระบบ การควบคุม และความสม่ำเสมอตลอดทั้งสายการผลิตมากกว่า
เมื่อใดที่การผลิตแบบเป็นชุดยังคงมีความเหมาะสมมากกว่าในอุตสาหกรรมยา?
การผลิตแบบเป็นชุดยังคงเหมาะสมเมื่อความยืดหยุ่น ความหลากหลายของผลิตภัณฑ์ หรือความไม่แน่นอนของโครงการ ทำให้การใช้สายการผลิตที่เชื่อมต่อกันอย่างแน่นหนาทำได้ยากขึ้น กระบวนการที่มีการแยกขั้นตอนมากขึ้นยังคงจัดการได้ง่ายกว่าเมื่อผลิตภัณฑ์มีการเปลี่ยนแปลงบ่อย เมื่อการพัฒนายังคงดำเนินต่อไป หรือเมื่อการบูรณาการสายการผลิตอย่างเต็มรูปแบบจะเพิ่มความซับซ้อนมากกว่าคุณค่า
การผลิตแบบเป็นชุดยังคงใช้งานได้จริงเมื่อกระบวนการจัดการหรือบรรจุภัณฑ์ในขั้นตอนถัดไปยังไม่พร้อมที่จะสอดคล้องกับจังหวะการผลิตในขั้นตอนต้นน้ำที่สม่ำเสมอกว่า หากการอัดเม็ด การตรวจสอบ หรือการบรรจุยังคงขึ้นอยู่กับการกำหนดตารางเวลาที่แบ่งเป็นส่วนๆ โครงสร้างการผลิตแบบเป็นชุดอาจเหมาะสมกับการดำเนินงานมากกว่า ดังนั้นรูปแบบที่เหมาะสมจึงขึ้นอยู่กับห่วงโซ่การผลิตทั้งหมด ไม่ใช่แค่กระบวนการในส่วนหน้าเท่านั้น

ผู้ผลิตยาควรตรวจสอบอะไรบ้างก่อนที่จะเปลี่ยนไปใช้กระบวนการผลิตแบบต่อเนื่อง?
ขั้นแรก ให้ถามว่าผลิตภัณฑ์และกระบวนการมีความเสถียรเพียงพอสำหรับสายการผลิตที่เชื่อมโยงกันมากขึ้นหรือไม่ การผลิตแบบต่อเนื่องจะให้ความสำคัญกับความสม่ำเสมอของวัตถุดิบ พฤติกรรมของวัสดุ และการประสานงานของการดำเนินงานมากขึ้น จุดอ่อนในขั้นตอนต้นน้ำจึงมีความสำคัญมากขึ้น
ประการที่สอง ให้ถามว่ากระบวนการอัดเม็ด การตรวจสอบ และการบรรจุหีบห่อ สอดคล้องกับจังหวะการผลิตที่สายการผลิตพยายามสร้างขึ้นหรือไม่ การเปลี่ยนไปใช้กระบวนการต้นน้ำที่เชื่อมโยงกันมากขึ้นโดยไม่ทบทวนการจัดการปลายน้ำ มีแนวโน้มที่จะสร้างปัญหาคอขวดใหม่ ไม่ใช่ระบบที่ดีขึ้น
ประการที่สาม ถามว่าทีมพร้อมที่จะจัดการการผลิตในฐานะระบบหรือไม่ การผลิตแบบต่อเนื่องต้องการการบูรณาการสายการผลิต ระบบอัตโนมัติ และกลยุทธ์การควบคุมที่มากกว่าเดิม มันให้ผลตอบแทนที่ดีกับการคิดแบบเชื่อมโยง แต่ก็เผยให้เห็นการวางแผนที่ไม่เชื่อมโยงกันอย่างรวดเร็วเช่นกัน
บทสรุป
ลองนึกถึงการผลิตแบบต่อเนื่องในอุตสาหกรรมยาว่าเป็นโครงสร้างการผลิตที่แตกต่างออกไป ไม่ใช่แค่เพียงวิธีการทำงานของเครื่องจักรที่เร็วขึ้นเท่านั้น มันเปลี่ยนวิธีการเคลื่อนย้ายวัสดุ วิธีที่กระบวนการแต่ละขั้นขึ้นอยู่กับกันและกัน และวิธีที่บรรจุภัณฑ์ปลายทางต้องสอดคล้องกับผลผลิตต้นทาง สำหรับผู้ผลิตยาเม็ด การถามคำถามที่แท้จริงไม่ใช่ว่าการผลิตแบบต่อเนื่องฟังดูทันสมัยหรือไม่ แต่เป็นว่าห่วงโซ่การผลิตและบรรจุภัณฑ์ทั้งหมดสามารถรองรับการไหลเวียนที่กระชับขึ้นซึ่งรูปแบบนี้ต้องการได้หรือไม่
คำถามที่พบบ่อย
การผลิตแบบต่อเนื่องในอุตสาหกรรมยาคืออะไร?
รูปแบบการผลิตที่วัสดุเคลื่อนย้ายผ่านกระบวนการที่เชื่อมโยงกันอย่างต่อเนื่อง แทนที่จะถูกจัดการเป็นชุดๆ แยกกันอย่างชัดเจน
การผลิตแบบต่อเนื่องแตกต่างจากการผลิตแบบเป็นชุดอย่างไร?
การผลิตแบบเป็นชุดจะมีการหยุด การขนย้าย และจุดพักระหว่างทางมากกว่า ในขณะที่การผลิตแบบต่อเนื่องจะช่วยลดช่วงหยุดพักเหล่านั้น และอาศัยการไหลเวียนที่ประสานงานกันตลอดทั้งสายการผลิตมากขึ้น
การผลิตแบบต่อเนื่องนั้นเกี่ยวข้องกับความเร็วที่สูงขึ้นเป็นหลักใช่หรือไม่?
ไม่ ความเร็วอาจเป็นผลลัพธ์อย่างหนึ่ง แต่การเปลี่ยนแปลงที่สำคัญกว่าคือโครงสร้างกระบวนการ โมเดลนี้เปลี่ยนจุดสนใจไปที่การป้อนวัตถุดิบที่เสถียร การประสานงานของสายการผลิต และจังหวะการผลิต
เหตุใดการผลิตแบบต่อเนื่องจึงส่งผลต่อบรรจุภัณฑ์?
เนื่องจากการไหลเวียนของวัตถุดิบต้นน้ำที่คงที่มากขึ้นจะเปลี่ยนแปลงวิธีการตรวจสอบ การจัดเก็บ และการบรรจุภัณฑ์ในขั้นตอนถัดไปให้สอดคล้องกับผลผลิต การวางแผนบรรจุภัณฑ์จึงเชื่อมโยงกับจังหวะการผลิตอย่างใกล้ชิดยิ่งขึ้น
การผลิตแบบต่อเนื่องสามารถทดแทนการผลิตแบบเป็นชุดได้เสมอหรือไม่?
ไม่ การผลิตแบบเป็นชุดอาจเหมาะสมกว่าในกรณีที่ความยืดหยุ่น ความหลากหลายของผลิตภัณฑ์ หรือข้อจำกัดในขั้นตอนถัดไปทำให้การบูรณาการสายการผลิตเต็มรูปแบบทำได้ยากขึ้น
ผู้ผลิตควรประเมินอะไรบ้างก่อนที่จะเปลี่ยนไปใช้การผลิตแบบต่อเนื่อง?
พิจารณาความเสถียรของวัตถุดิบ การบูรณาการกระบวนการ การอัดเม็ดยา และการจับคู่บรรจุภัณฑ์ รวมถึงพิจารณาว่าสายการผลิตทั้งหมดสามารถรองรับการไหลเวียนของการผลิตที่เชื่อมโยงกันมากขึ้นได้หรือไม่
เอกสารอ้างอิง
●
องค์การอาหารและยา (FDA) มาตรา 13 การผลิตสารออกฤทธิ์และผลิตภัณฑ์ยาอย่างต่อเนื่อง (
เอฟดีเอฟ
)
●
ISPE, การผลิตยาเม็ดรับประทานแบบต่อเนื่อง (
ไอเอสพี
)































 รองรับเครือข่าย IPv6
รองรับเครือข่าย IPv6